SPR imaging of photo-cross-linked small molecule microarrays on gold.
Outlines:
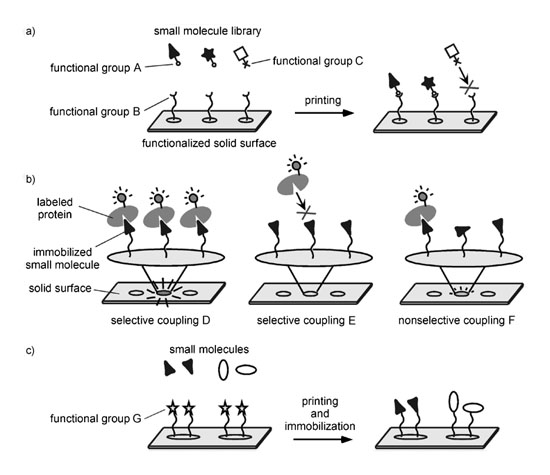
We have recently developed a "photoaffinity" surface plasmon resonance (SPR) imaging platform to enable the immobilization of structurally diverse small molecules on solid supports in a "functional group-independent" manner.
A gold-coated glass chip (TOYOBO, Japan) was immersed in the ethanol solution, which contained 0.1 mM photo-cross-linker and 0.9 mM dummy linker, for 16 h. The chip was rinsed successively with ethanol, water, and ethanol and dried under an air stream to obtain a photoreactive chip. DMSO solutions (10 mM) of small molecules were spotted on the chip using an automated spotter (TOYOBO, Japan). The obtained chip was dried in vacuo to evaporate the DMSO, and the chip was then irradiated under a UV transmission filter (Sigma-Koki, Japan) with a Stratalinker 1800 UV cross-linker (Stratagene, CA) or a CL-1000L ultraviolet cross-linker (UVP Inc., CA). The irradiated energy on the chip amounted to 2.8 J/cm2. The surface was repeatedly rinsed with DMSO, water, and ethanol.
The photo-cross-linked small-molecule array was placed in an SPR imaging instrument (TOYOBO, Japan). The SPR signals were obtained in the running buffer (10 mM HEPES, 150 mM NaCl, pH 7.2). The running buffer and the protein samples in the same buffer were applied to the array surface at 100 uL/min. All SPR experiments were performed at 30 oC. The SPR image and signal data were collected with an SPR analysis program (TOYOBO, Japan). The SPR difference image was constructed by using Scion Image (Scion, MD).
 |
|
Overview of small-molecule microarrays. |
References:
-
K. Kawai, A. Saito, T. Sudo, H. Osada.
Specific Regulation of Cytokine-Dependent p38 MAP Kinase Activation by p625/SQSTM1.
J. Biochem., 143, 763-772 (2008). -
A. Saito, K. Kawai, H. Takayama, T. Sudo, H. Osada.
Improvement of photoaffinity SPR imaging platform and determination of the binding site of p62/SQSTM1 to p38 MAP kinase.
Chem. Asian J., 3, 1607-1612 (2008). -
N. Kanoh, M. Kyo, K. Inamori, A. Ando, A. Asami, A. Nakao, H. Osada.
SPR imaging of photo-cross-linked small molecule microarrays on gold.
Annal. Chem., 78, 2226-2230 (2006).


