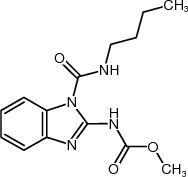
- InChI=1S/C14H18N4O3/c1-3-4-9-15-13(19)18-11-8-6-5-7-10(11)16-12(18)17-14(20)21-2/h5-8H,3-4,9H2,1-2H3,(H,15,19)(H,16,17,20)
- InChIKey=RIOXQFHNBCKOKP-UHFFFAOYSA-N
- 17804-35-2
- Benomyl
- Therapeutic
-
Antifungal
- Target
-
β-Tubulin
Inhibition of microtubule polymerization of bovine brain tubulin in vitro (IC50: 58.3 μM)
Inhibition of HeLa cell proliferetion (IC50: 5 μM) and the cell cycle progression during mitosis (IC50: ca 15 μM)
-
Gupta K, Bishop J, Peck A, Brown J, Wilson L, Panda D
Antimitotic antifungal compound benomyl inhibits brain microtubule polymerization and dynamics and cancer cell proliferation at mitosis, by binding to a novel site in tubulin.
Biochemistry, 43(21): 6645-6655 (2004) 15157098 doi: 10.1021/bi036112v
15157098 doi: 10.1021/bi036112v -
Friedman PA, Platzer EG
Interaction of anthelmintic benzimidazoles and benzimidazole derivatives with bovine brain tubulin.
Biochim Biophys Acta, 544(3): 605-614 (1978) 728472
728472 -
Hein L Klopping
(1971). Carbamoyl substituted 2-aminobenzimidazoles. US3631176. -
G. J. Bollen, A. Fuchs
On the specificity of the in vitro and in vivo antifungal activity of benomyl
Netherlands Journal of Plant Pathology, 76(6): 299-312 (1970) doi: 10.1007/BF03041361
