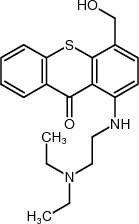
- InChI=1S/C20H24N2O2S/c1-3-22(4-2)12-11-21-16-10-9-14(13-23)20-18(16)19(24)15-7-5-6-8-17(15)25-20/h5-10,21,23H,3-4,11-13H2,1-2H3
- InChIKey=MFZWMTSUNYWVBU-UHFFFAOYSA-N
- 3105-97-3
- Hycanthone
- Therapeutic
-
Anticancer
Antischistosomiasis
- Target
-
Apurinic endonuclease-1 (APE1)
Antitumor activity for P388 lymphocytic leukemia-bearing mice
Inhibition of APE1 endonuclease activity by direct binding to the hydrophobic site (Kd = 10 nM)
-
Naidu MD, Agarwal R, Pena LA, Cunha L, Mezei M, Shen M, Wilson DM 3rd, Liu Y, Sanchez Z, Chaudhary P, Wilson SH, Waring MJ
Lucanthone and its derivative hycanthone inhibit apurinic endonuclease-1 (APE1) by direct protein binding.
PLoS One, 6(9): e23679 (2011) 21935361 doi: 10.1371/journal.pone.0023679
21935361 doi: 10.1371/journal.pone.0023679 -
Archer S, Pica-Mattoccia L, Cioli D, Seyed-Mozaffari A, Zayed AH
Preparation and antischistosomal and antitumor activity of hycanthone and some of its congeners. Evidence for the mode of action of hycanthone.
J Med Chem, 31(1): 254-260 (1988) 3336024
3336024 -
Rosi D, Peruzzotti G, Dennis EW, Berberian DA, Freele H, Archer S
A new active metabolite of "Miracil D".
Nature, 208(5014): 1005-1006 (1965) 4958061
4958061
