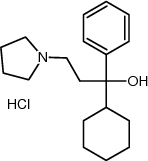
- InChI=1S/C19H29NO.ClH/c21-19(17-9-3-1-4-10-17,18-11-5-2-6-12-18)13-16-20-14-7-8-15-20;/h1,3-4,9-10,18,21H,2,5-8,11-16H2;1H
- InChIKey=ZFSPFXJSEHCTTR-UHFFFAOYSA-N
- 1508-76-5
- Procyclidine hydrochloride
- Therapeutic
-
Anticholinergic
Antiparkinsonian
- Target
-
Muscarinic acetylcholine receptor M1, M2, and M4
(S)-Procyclidine had a lower affinity than (R)-procyclidine
-
Waelbroeck M, Camus J, Tastenoy M, Lambrecht G, Mutschler E, Tacke R, Christophe J
Stereoselectivity of procyclidine binding to muscarinic receptor subtypes M1, M2 and M4.
Eur J Pharmacol, 189(2-3): 135-142 (1990) 2253700
2253700 -
DUFFIN WM, GREEN AF
The pharmacological properties of the optical isomers of benzhexol, procyclidine, tricyclamol and related compounds.
Br J Pharmacol Chemother, 10(3): 383-386 (1955) 13269719
13269719 -
Wilkinson Samuel, Adamson Donald Wallace
(1954). Catalytic reduction of diphenyl alkanolamines and resulting products. US2682543.
