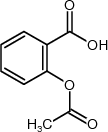
- InChI=1S/C9H8O4/c1-6(10)13-8-5-3-2-4-7(8)9(11)12/h2-5H,1H3,(H,11,12)
- InChIKey=BSYNRYMUTXBXSQ-UHFFFAOYSA-N
- 50-78-2
- Aspirin
- Therapeutic
-
Analgesic and antipyretic
Antiinflammatory
Antiplatelet
- Target
-
Cyclooxygenase
Growth inhibition against various cancer cell linces at sub-mM range
-
Mahdi JG, Mahdi AJ, Mahdi AJ, Bowen ID
The historical analysis of aspirin discovery, its relation to the willow tree and antiproliferative and anticancer potential.
Cell Prolif, 39(2): 147-155 (2006) 16542349 doi: 10.1111/j.1365-2184.2006.00377.x
16542349 doi: 10.1111/j.1365-2184.2006.00377.x -
Mitchell JA, Akarasereenont P, Thiemermann C, Flower RJ, Vane JR
Selectivity of nonsteroidal antiinflammatory drugs as inhibitors of constitutive and inducible cyclooxygenase.
Proc Natl Acad Sci U S A, 90(24): 11693-11697 (1993) 8265610
8265610 -
H. Dreser
Pharmakologisches über Aspirin (Acetylsalicylsäure)
Pflugers Arch, 76(5-6): 306-318 (1899) doi: 10.1007/BF01662127 -
T.J. Maclagan
THE TREATMENT OF ACUTE RHEUMATISM BY SALICIN AND SALICYLIC ACID
Lancet, 4: 342-383 (1876) doi: 10.1016/S0140-6736(02)46061-8
