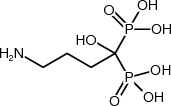
- InChI=1S/C4H13NO7P2/c5-3-1-2-4(6,13(7,8)9)14(10,11)12/h6H,1-3,5H2,(H2,7,8,9)(H2,10,11,12)
- InChIKey=OGSPWJRAVKPPFI-UHFFFAOYSA-N
- 66376-36-1
- Alendronate
- Therapeutic
-
Malignant
Paget's disease bone
Postmenopausal osteoporosis
- Target
-
Farnesyl pyrophosphate synthase (FPPS)
Inhibition of the incorporation of [3H]-mevalonolactone both into prenylated proteins and into nonsaponifiable lipids in osteoclasts (IC50 = 15 μM)
Prevention of early trabecular bone loss and cartilage degeneration following non-invasive knee injury using mice assay (1000 μg/kg/dose as high-dose alendronate treatment )
Increase in sensitivity of Huh-7 HCC cell against the drug-combination (5 μM alendronate + 30 μM 5'-azacytizine)
-
Ilyas A, Hashim Z, Zarina S
Effects of 5'-azacytidine and alendronate on a hepatocellular carcinoma cell line: a proteomics perspective.
Mol Cell Biochem, 405(1-2): 53-61 (2015) 25854900 doi: 10.1007/s11010-015-2395-1
25854900 doi: 10.1007/s11010-015-2395-1 -
Khorasani MS, Diko S, Hsia AW, Anderson MJ, Genetos DC, Haudenschild DR, Christiansen BA
Effect of alendronate on post-traumatic osteoarthritis induced by anterior cruciate ligament rupture in mice.
Arthritis Res Ther, 17: 30 (2015) 25888819 doi: 10.1186/s13075-015-0546-0
25888819 doi: 10.1186/s13075-015-0546-0 -
Christiansen BA, Anderson MJ, Lee CA, Williams JC, Yik JH, Haudenschild DR
Musculoskeletal changes following non-invasive knee injury using a novel mouse model of post-traumatic osteoarthritis.
Osteoarthritis Cartilage, 20(7): 773-782 (2012) 22531459 doi: 10.1016/j.joca.2012.04.014
22531459 doi: 10.1016/j.joca.2012.04.014 -
Bergstrom JD, Bostedor RG, Masarachia PJ, Reszka AA, Rodan G
Alendronate is a specific, nanomolar inhibitor of farnesyl diphosphate synthase.
Arch Biochem Biophys, 373(1): 231-241 (2000) 10620343 doi: 10.1006/abbi.1999.1502
10620343 doi: 10.1006/abbi.1999.1502 -
van Beek E, Pieterman E, Cohen L, Löwik C, Papapoulos S
Farnesyl pyrophosphate synthase is the molecular target of nitrogen-containing bisphosphonates.
Biochem Biophys Res Commun, 264(1): 108-111 (1999) 10527849 doi: 10.1006/bbrc.1999.1499
10527849 doi: 10.1006/bbrc.1999.1499 -
Helmut Blum, Karl-Heinz Worms
(1983). Process for the production of ω-amino-1-hydroxyalkylidene-1,1-bisphosphonic acid. US4407761 A.
