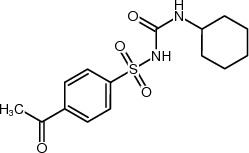
- InChI=1S/C15H20N2O4S/c1-11(18)12-7-9-14(10-8-12)22(20,21)17-15(19)16-13-5-3-2-4-6-13/h7-10,13H,2-6H2,1H3,(H2,16,17,19)
- InChIKey=VGZSUPCWNCWDAN-UHFFFAOYSA-N
- 968-81-0
- Acetohexamide
- Dymelor
- Therapeutic
-
Antidiabetes (diabetes mellitus type 2)
Hypoglycemic agent
- Target
-
ATP-sensitive inward rectifier potassium channel 11 (Sulfonylurea receptor 1, Kir 6.2)
Inhibitor of [3H]glibenclamide (IC50 = 330 μM)
Binding to human serum albumin (HSA) with high affinity binding site (Ka = 1.3 (± 0.2) × 105 M-1) and low binding site (Ka = 3.5 (± 3.0) × 102 M-1)
-
Rehmann H
Epac2: a sulfonylurea receptor?
Biochem Soc Trans, 40(1): 6-10 (2012) 22260657 doi: 10.1042/BST20110640
22260657 doi: 10.1042/BST20110640 -
Joseph KS, Hage DS
Characterization of the binding of sulfonylurea drugs to HSA by high-performance affinity chromatography.
J Chromatogr B Analyt Technol Biomed Life Sci, 878(19): 1590-1598 (2010) 20435530 doi: 10.1016/j.jchromb.2010.04.019
20435530 doi: 10.1016/j.jchromb.2010.04.019 -
Miki T, Nagashima K, Seino S
The structure and function of the ATP-sensitive K+ channel in insulin-secreting pancreatic beta-cells.
J Mol Endocrinol, 22(2): 113-123 (1999) 10194514
10194514 -
Inoue Y, Emoto M, Inoue H, Kaku K, Kaneko T
Characterization of the binding sites for [3H]glibenclamide in rat liver membranes.
Eur J Pharmacol, 284(1-2): 77-82 (1995) 8549639
8549639 -
MARSHALL FJ, SIGAL MV Jr, SULLIVAN HR, CESNIK C, ROOT MA
FURTHER STUDIES ON N-ARYLSULFONYL-N-ALKYLUREAS.
J Med Chem, 6: 60-63 (1963) 14175624
14175624
